 StatStrip® Lactate and StatStrip Xpress® Lactate Systems
StatStrip® Lactate and StatStrip Xpress® Lactate Systems
Features:
- Fastest turnaround – 13 seconds
- 0.6 μL whole blood sample
- Excellent correlation to central laboratory reference methods
The StatStrip Lactate Hospital Meter System is a handheld point-of-care testing system that brings lactate testing directly to the patient’s bedside.
Lactate is currently measured on blood gas analyzers, which creates numerous problems for bedside testing. Blood gas analyzers require anaerobic arterial samples, large sample volumes (100-200 microliters), and long analysis times (up to 2.5 minutes).
In addition, blood gas analyzers are complex to operate, fixed in location, and expensive to purchase and use for serial lactate testing.
StatStrip Lactate provides the fastest turnaround time possible (13 seconds), on the smallest whole blood sample (0.6 microliters), and can be easily operated by medical and nursing staff. StatStrip Lactate is a very low-cost device that makes lactate testing practical and affordable in any size ED, ICU, or medical unit. It is fully connectable to a hospital's IT system through NovaNet.
For more product information, click here or contact Nova Biomedical at:
Nova Biomedical / 200 Prospect Street / Waltham, MA 02454 / 781-894-0800
For Nova Biomedical Sales inquiries, reach us at 1-800-458-5813.
For Technical Support, reach us at 1-800-545-NOVA (6682).
To place an order, call 1-800-822-0911 or email novaorders@novabio.com
Specifications
StatStrip Lactate Hospital Meter System
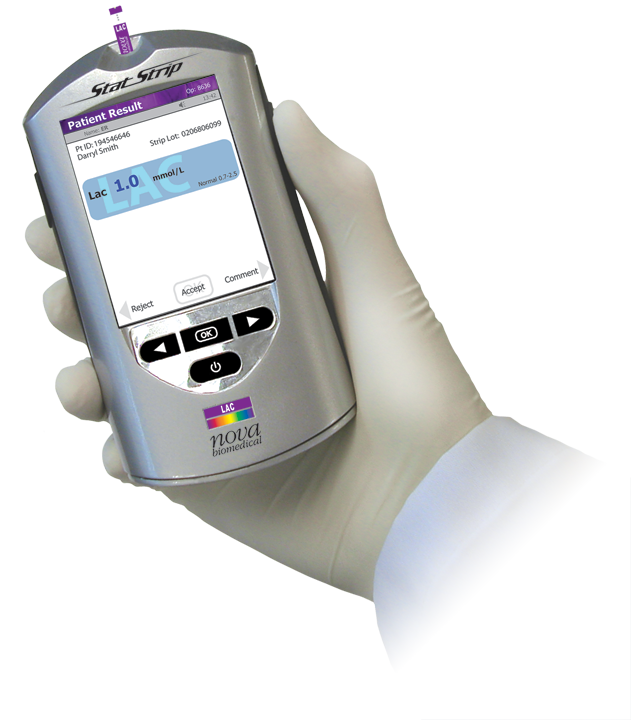
Weight: 0.49 lb (220 g)
Size: 5.8 in x 3.1 in x 1.18 in
(147 mm x 79 mm x 30 mm)
Data Storage:
Patient Tests: 1,000 tests
QC Tests: 200 tests
Users: 8,000 users
Connectivity:
Meter Data Output: RJ-45 Ethernet Port
Protocol: TCP/IP Ethernet 100 Mbit
Standard: POCT1-A2 Compliant
Setup Program: NovaNet™ Web-Based Instrument Manager Software
Battery Information:
Type: 3.7V Li Polymer Rechargeable Battery
Features: Rechargeable/Replaceable
Life: 6-8 hours in use (approximately 40 tests w/barcode scans)/12-24 hours standby
Docking Station:
Optional accessory; desk or wall mount charging station with RJ-45 Ethernet jack. 3 LED indicator lights showing connection status, transmit/receive data, and charging status. Extra battery slot included for recharging and storage of spare battery.
Additional Features:
- Color touchscreen
- Built-in barcode scanner
- Traditional QC with target values assigned to QC materials
- Numeric or pass/fail QC
- QC prompting or QC lock out
- Abnormal and critical ranges flagged by color highlighting
- Canned or free text user comments
- Rejecting of results at meter (optional) for repeat testing
- Accepting patient ID or accession number
- Use of operator password (lockout)
- Entry of offline tests
- Entry of ICD-9 Diagnosis codes
- Ordering physicians ID
- Onscreen training mode
- Correlation mode
- Slope and intercept adjustment
- Web-based meter configuration by location
- Custom, web-based meter configuration by location
- Connect to hospital ADT
- Connect to all LIS/EMR
- POCT1-A2 data output
StatStrip Xpress Lactate Meter
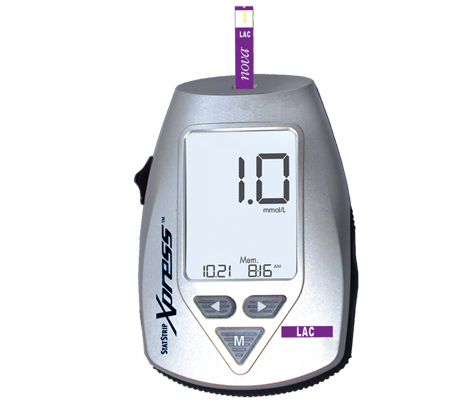
StatStrip Xpress Lactate not yet available in the U.S. and Canada
Designed for applications that do not need data interfacing and connectivity, Xpress Lactate retains all of the analytical performance features of StatStrip Lactate.
Weight: 0.2 lb (75 g)
Size: 3.6 in x 2.3 in x 0.9 in (91 mm x 58 mm x 23 mm)
Data Storage:
Patient & QC Tests: 400 tests total (FIFO)
Connectivity:
Data Transfer: Strip Port Connection to USB
Data Program: Nova Microsoft-Excel based data transfer software
Battery Information:
Type: 3V Li Button Battery
Features: Replaceable
Life: Minimum 600 tests
Additional Features:
- LCD black/white display
- Large numeric display (30 mm)
- Traditional QC with target values assigned to QC materials
- Units of measure based on meter ordered (mg/dl or mmol/L models)
- Automatic shut-off when not in use
- Automatic sample detection and analysis start
- Automatic sample counter with date/time stamp for data tracking
StatStrip Lactate Test Strips
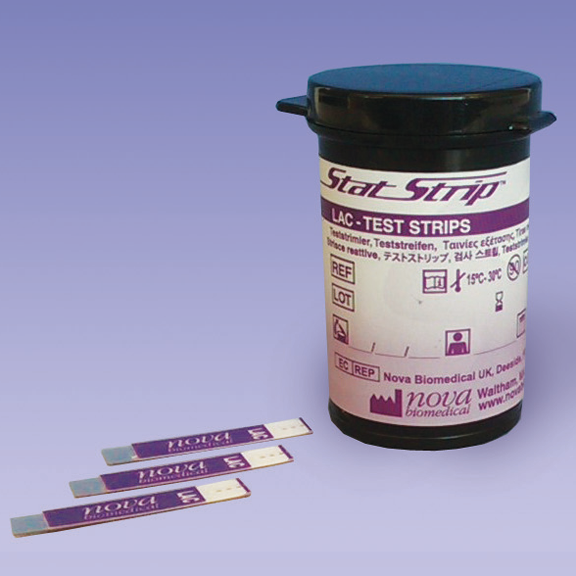
Tests:
Tests Measured: Blood Lactate, Hematocrit Corrected
Test Reported: Lactate
Test Time: 13 Seconds
Test Strip Volume: 0.6 µL
Test Methodology: Electrochemistry
Sample Types & Operating Modes:
Whole Blood: Arterial, Venous
Lactate Measurement Range:
0.3-20.0 mmol/L (3-180 mg/dL)
No Interferences From:
Hematocrit, Ascorbic Acid, Uric Acid, Acetaminophen (Paracetamol), Bilirubin
Operating Ranges:
Temperature: 59°F-104°F (15°C- 40°C)
Altitude: Up to 15,000 feet
Humidity: 10% to 90% relative humidity
Hematocrit: 20% – 65%
Reagents and Strips:
Strips: 25 strips per vial; 2 vials per package
QC: Three levels (Low, Normal, High);
sold separately
Linearity: Five levels available
Test Strip & QC Stability:
24 months from date of manufacturing
z
3 months open-vial stability
Certifications and Compliance: Nova Biomedical complies with the FDA Quality Systems Regulations and is certified to EN ISO 13485:2016. Complies to IVDD. Tested according to: EN 61010-1:2010, EN 61010-2-101:2015, EN 60825-1/A1:2014. Certifications and compliances applicable in countries where regulations require.
Specifications current as of revision date.