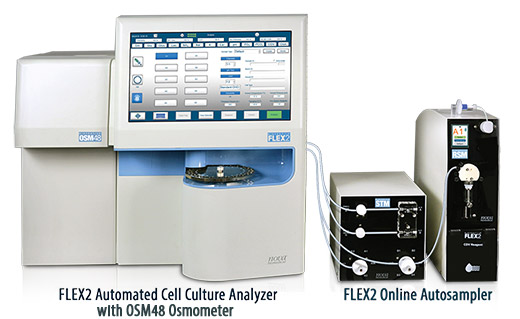
BioProfile® FLEX2
BioProfile® FLEX2
Automated Cell Culture Analyzer with Maintenance-Free Chemistry Sensors
BioProfile FLEX2 combines Nova’s groundbreaking MicroSensor™ Card technology with optical measurement and freezing point osmometry for an automated and comprehensive cell culture analyzer that eliminates chemistry sensor maintenance, increases analyzer speed, and reduces sample volume. The full 16 cell culture test menu includes: Gluc, Lac, Gln, Glu, NH4+, Na+, K+, Ca++, pH, PCO2, PO2, total cell density, viable cell density, viability, cell diameter, and osmolality.
Compared to the previous generation BioProfile FLEX there is no maintenance for any of the 11 chemistry and gas sensors, sample volume for the full test menu is reduced by 75% to 275 microliters, and testing time is reduced to just 4.5 minutes. Automated sampling from 96-well plates, syringes, or a 24-position external “load-and-go” sample tray provides maximum workflow flexibility and efficiency for cell culture monitoring.
Fully Automated Sampling Options
FLEX2 Online Autosampler
BioProfile FLEX2 is capable of automated online sampling. Nova’s FLEX2 On-Line Autosampler (OLS) provides fully automated sampling from virtually all culture systems from single-use bench scale bioreactors to large production bioreactors. When connected to an OPC-compatible control system, FLEX2 with OLS provides real-time data transmission for automated sampling, analysis, and feedback control of all measured parameters. The OLS can connect up to 10 vessels and setup time for a full bench of 10-bioreactors can be completed in less than 20 minutes.
FLEX2 and ESM integration with ambr® 250 and ambr 15 cell culture systems
Nova has also integrated the FLEX2 analyzer with ambr 15 and 250 microbioreactors to provide a fully automated system. FLEX2’s optional External Sampling Module (ESM) automatically delivers cell culture samples from ambr® 250 and ambr® 15 microbioreactor systems to FLEX2 for analysis.

Maintenance-free microsensor cards 
Chemistry and gas biosensors are combined in credit-card-sized MicroSensor cards, which use proven Nova biosensor technology that has been validated in thousands of cell culture processes. MicroSensor Cards are maintenance-free, and replaceable in seconds.
Cartridge-based reagent management system (RMS)
FLEX2’s RMS features smart reagent cartridges that are easy to use with simple snap-in replacement.• RMS automatically captures cartridge installation date and time, lot number, and expiration.
• RMS monitors reagent usage and provides real-time reagent status and alerts, maximizing analyzer readiness.
• A self-contained waste receptacle within the cartridge eliminates direct handling of waste and direct contact with hazardous trypan blue and biological materials. Other systems can pose significant waste handling hazards to operators.
Fast analysis time
Comprehensive test results including cell density/viability, pH/gases, and key chemistries are available in 4.5 minutes. Throughput for individual modules is 2.0 minutes.Small sample volume
Sample volume is 275 μL for a full 16-test profile, enabling comprehensive testing even from low volume culture systems. Individual modules require as little as 135 μL.Analysis time and sample size when modules are run individually
| Module | Analysis Time | Sample Size |
Gas Module: |
120 sec | 275 μl |
| Chemistry Module: Gluc, Lac, Gln, Glu, NH4+, Na+, K+, Ca++ |
120 sec | 135 μl |
| Osmometry Module: Osmolality |
240 sec | 145 μl |
| Cell Density/Viability Module: Total cell density, viability, cell diameter |
240 sec | 135 μl |
Analysis time and sample size when all modules are run collectively
| All Modules-All Tests | 270 sec | 275 μl |
 Walkaway automation
Walkaway automation
FLEX2’s high capacity, 48-position osmometer enables walkaway, automated sampling of up to 48 samples without any user intervention, significantly prolonging maintenance intervals. The high capacity tray also enables completely automated sampling for all tests, including osmometry, for automated culture systems such as the Sartorius ambr®15. This eliminates the need to replace osmometer tubes in the middle of a sampling set and allows for sample scheduling in the middle of the night, on weekends, and holidays, without the need to perform any maintenance during the full batch of automated samples. This capability frees the instrument to perform regular sampling of non-automated cultures during normal business hours.
Maximum Workflow Efficiency
Simple touchscreen operation, a choice of three sampling modes, fast analysis time, and automated quality control (QC) provide maximum simplicity, labor savings, and workflow efficiency for cell culture monitoring.Onboard automated QC
Onboard liquid QC provides true verification of FLEX2 performance and saves hours of labor each week compared to manual QC testing. QC cartridges contain a 30-day supply of QC material. Controls are run automatically at user-selected intervals and QC data is automatically logged and filed in a variety of formats.
24-position, external load-and-go tray
 An external load-and-go sample tray allows for continuous loading of cell culture samples for the most flexible and efficient sample throughput.
An external load-and-go sample tray allows for continuous loading of cell culture samples for the most flexible and efficient sample throughput.
Automated, 96-well plate analysis
 FLEX2 is the only cell culture analyzer to offer automated sampling from 96-well plates.
FLEX2 is the only cell culture analyzer to offer automated sampling from 96-well plates.
Syringe or cup sampling
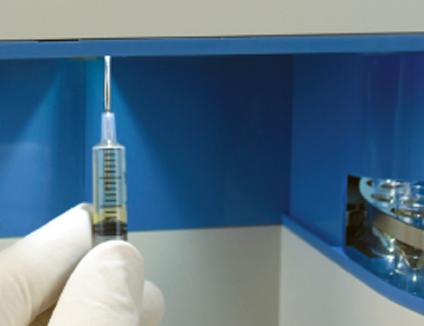 Individual samples also can be analyzed directly from syringes or cups. Batch sampling via the 96-well plate and/or external “load-and-go” sample tray runs can be interrupted at any time to run critical gas syringe samples.
Individual samples also can be analyzed directly from syringes or cups. Batch sampling via the 96-well plate and/or external “load-and-go” sample tray runs can be interrupted at any time to run critical gas syringe samples.
Advanced OPC Connectivity
Nova’s OPC connectivity integrates FLEX2 with any OPC-compliant devices, such as bioreactor controllers, data historians, laboratory information management systems (LIMS), and plant management systems. Nova’s OPC connectivity features:- Automated bidirectional data and control commands
- Data archiving
- Connection to any OPC-compliant device
- Connectivity verification
- Bioreactor feedback control
- Remote monitoring of status and data
GMP compliance
FLEX2 meets GMP manufacturing requirements through installation qualification (IQ) and operational qualification (OQ) documentation and validation support from Nova specialists.Compliance with 21 CFR Part 11
Limited accessUser log-on is secured by both user ID and password. Automatic log-off features prevent unauthorized access.
Electronic record retention and retrieval
- All data are securely retained through password access control in both human-readable and electronic forms.
- Records are readily retrievable throughout their retention period.
Audit trails
- Time-stamped audit trails record the date and time of operator entries and actions that create, modify, or delete electronic records.
- Record changes do not obscure previously recorded information.
- Records are maintained in original and audited form.
For more product information, click here or contact Nova Biomedical at:
Nova Biomedical / 200 Prospect Street / Waltham, MA 02454 / 781-894-0800
For Nova Biomedical Sales inquiries, reach us at 1-800-458-5813.
For Technical Support, reach us at 1-800-545-NOVA (6682).
To place an order, call 1-800-822-0911 or email novaorders@novabio.com